Waist Cincher vs Waist Lipo: Which is better to address post baby belly
Waist cinchers are dangerous. There’s a better, safer & longer-lasting way to get the waist you’ve always wanted! Waist lipo can address post baby belly.
Those in Singapore searching for non-surgical skin lift may come across two names: HIFU and Ultherapy®. They are used interchangeably, but they are not the same treatment.
Unlike standard HIFU, Ultherapy® uses Micro-Focused Ultrasound technology with real-time imaging, which allows the doctor to visualise the deep tissue layers and deliver energy precisely where it is needed.
At Amaris B. Clinic, the treatment offered is Ultherapy® Prime, a modern generation of this technology, featuring enhanced DeepSEE® imaging and faster processing for more accurate, effective results.
It targets the foundational SMAS layer at a depth of 4.5mm, the same layer addressed in a surgical facelift, to stimulate collagen regrowth.
Over two to three months, skin gradually lifts and tightens from within. Treatment areas include the face, brows, neck, under the chin, and décolletage.
Only FDA-cleared, authentic Ultherapy® Prime machines are used. All treatments are performed personally by Dr Ivan Puah.
If you're looking for noticeable skin lifting and tightening without surgery or downtime, Ultherapy is your premier solution.

Ultherapy® uses Micro-Focused Ultrasound (MFU) energy to gently target the deep structural layers of your skin—the same layers addressed in a surgical facelift.
What makes it truly advanced is its integrated imaging technology. As the doctor glides the device over your skin, he can see the tissue layers in real-time, ensuring energy is delivered exactly where it’s needed for optimal results.
The ultrasound energy penetrates safely to a depth of up to 5mm, creating heat that triggers your body’s natural regenerative process.
This jumpstarts neocollagenesis — your body’s production of fresh, new collagen. Over time, this new collagen gradually lifts and tightens loose skin from within, for a result that looks completely natural.

| Treatments | HIFU | THREADLIFT | ULTHERAPY PRIME |
|---|---|---|---|
| FDA approved | Varies by device | Yes (PDO threads) | Yes |
| Real-time Imaging | No | No | Yes (DeepSEE) |
| Treatment Depth | 3mm – 4.5mm | Subcutaneous | Up to 4.5mm (SMAS) |
| Results Duration | 6 – 12 months | 12 – 18 months | 12 – 18 months |
| Downtime | None | 1 – 2 weeks | None |
| Invasiveness | Non-invasive | Minimally invasive | Non-invasive |
| Best for | Budget-conscious, mild skin laxity | Mild to moderate skin laxity | Mild-moderate laxity, precision lifting |
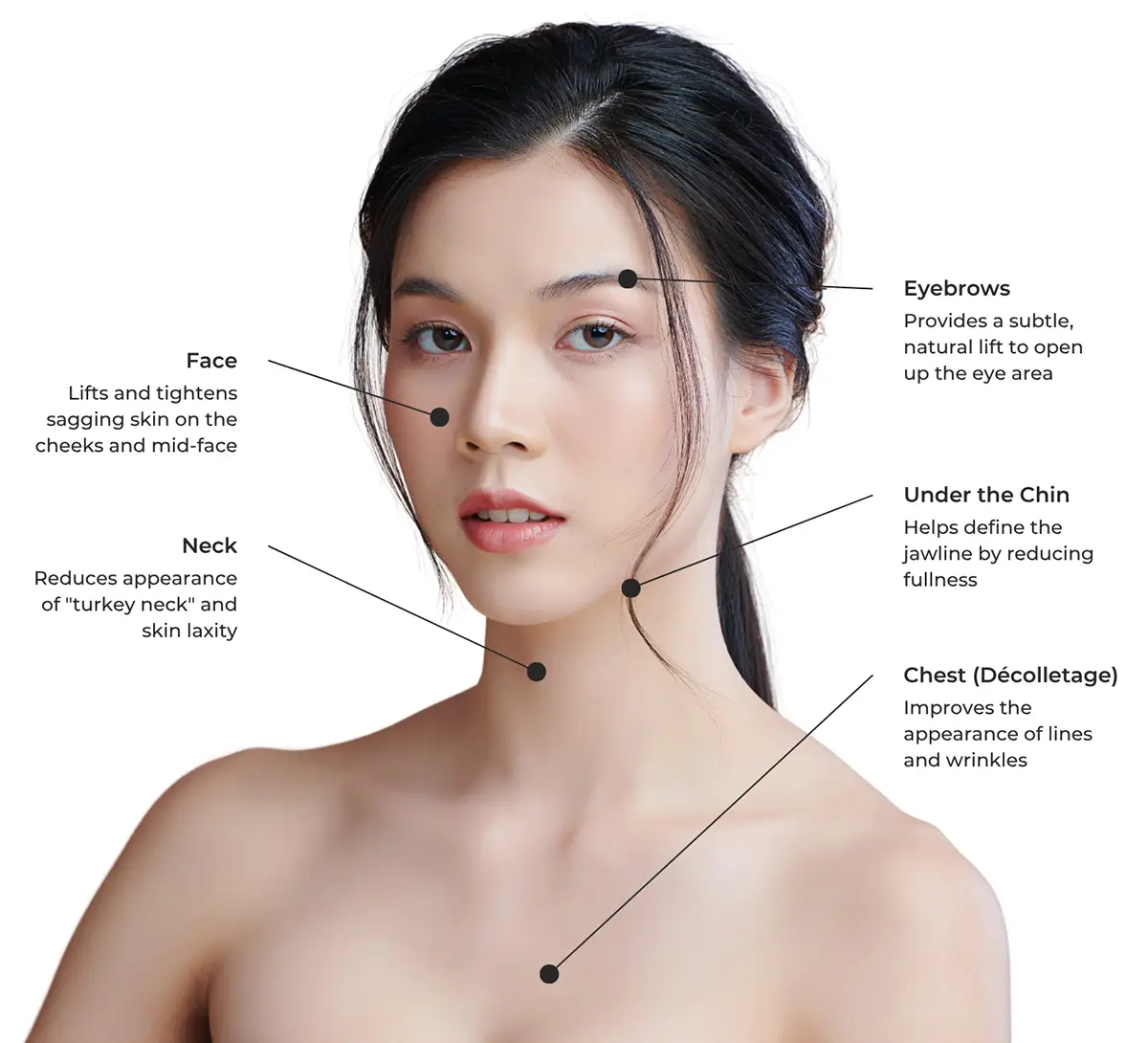
Ultherapy® is very versatile and is FDA-approved for lifting and tightening some of the most common areas of facial and skin ageing concerns.
Ultherapy® is the first and only FDA-approved non-invasive treatment for lifting the eyebrows, under the chin, and chest.
At Amaris B. Clinic, we are proud to be a certified provider of authentic Ultherapy® in Singapore. Our team is committed to carefully curating medical technologies that are proven, effective, and truly beneficial for our patients. It is this philosophy that guides our introduction of Ultherapy® Prime—the latest evolution in non-surgical skin lifting.
Building on two decades of innovation since the first-generation device, Ultherapy® Prime represents the advancement in focused ultrasound technology. We believe it offers our patients an even more refined experience and effective solution for achieving a naturally lifted and firmer appearance without surgery or downtime.
Ultherapy® Prime features an optimised design specifically for delicate areas like the brow and under the chin.
Ultherapy® Prime stands out due to its advanced DeepSEE® technology. The enhanced real-time imaging allows our doctor to visualise skin layers during treatment, ensuring precise energy delivery to the foundational SMAS layer at 4.5mm depth – the same layer targeted in surgical facelifts.
The key difference? Instead of cutting, this energy gently stimulates your skin’s natural regenerative process, kickstarting the production of collagen and elastin. These are the essential proteins responsible for your skin's youthful strength and elasticity.
Provides clearer, sharper real-time imaging of tissue layers for unparalleled precision
Delivers immediate response times and more pronounced lifting effects
Shorter treatment sessions without compromising results
Most clients achieve noticeable improvement after just one treatment
Reaches where it matters most for natural-looking lifting
See initial lifting right away, with continued improvement over 2 to 3 months
Ultherapy® Prime is great for those who are starting to see mild to moderate signs of ageing but are not ready for surgery. It is not a substitute for a surgical facelift, but an excellent non-surgical alternative for lifting and tightening.
You may be a good candidate if you have:
The treatment is safe for all skin types and is most effective for individuals typically between their 30s and 60s, when collagen production naturally begins to slow.
At Amaris B. Clinic, we believe that clarity reduces anxiety.
That’s why your Ultherapy® Prime treatment is guided through a structured journey — from your first consultation to post-treatment care — so you always know what to expect.
Your journey starts with a comprehensive medical history review to determine if you are a suitable candidate. This one-on-one session with Dr Ivan Puah focuses on personalising your treatment plan.
Whether you're targeting sagging brows, a double chin, jowls, or overall facial laxity, your Ultherapy® Prime session will be tailored to the areas that matter most to you.
We'll also discuss expected outcomes, how long results may last, and realistic timelines.
Ultherapy® Prime is a non-invasive ultrasound skin lifting procedure that typically takes 45 to 90 minutes, depending on the treatment area.
Here’s what happens:
You may feel heat or tingling during the procedure, which is normal and temporary. There is no downtime, and you can return to daily activities immediately after.
Return to your usual activities immediately.
Some patients may notice obvious positive changes after only a week.
Collagen production level increased tremendously. Your skin is more lifted and appears more youthful.
Results continue to improve. Skin will feel tighter and bouncier.
Most patients see initial skin tightening effects, but full results develop gradually over 2 to 3 months as collagen regenerates.
Aftercare includes:

Amaris B. Clinic has chosen to offer Ultherapy® Prime because its ability to deliver effective, safe, and convenient results aligns perfectly with our commitment to providing thoughtfully curated technologies that genuinely benefit our patients.
No incisions, needles, or scalpels. It’s a safe, gentle alternative to a surgical facelift.
Return to your daily routine immediately after your session.
Your skin lifts and tightens over 2 to 3 months as your body produces new collagen, for a refreshed look that is never overdone.
The ultrasound technology allows our doctor to see and treat the deep structural layers of your skin with unmatched accuracy.
Effectively addresses sagging skin on the face, neck, and décolletage (chest).
Backed by FDA-clearance, offering you a peace of mind.
Improve the appearance of lines and wrinkles on the décolletage.
Lift and tighten the skin naturally on your brows, neck, and under the chin.
Amaris B. Clinic uses only FDA-certified Ultherapy® Prime machines, guaranteeing an authentic and safe treatment experience. You will receive clear, honest guidance from consultation to pricing — no surprises, just professional care backed by over 20 years of medical excellence.
The cost of an Ultherapy® Prime procedure in Singapore is based on the areas to be treated. Please schedule a consultation appointment for more accurate pricing information.
Your skin lifting results depend greatly on the expertise and experience of your provider. At Amaris B. Clinic, your treatment is personally performed by Dr Ivan Puah. With his technique and deep understanding of facial anatomy, Dr Puah ensures the ultrasound energy is delivered accurately for optimal lifting and tightening.
Is Ultherapy safe?
Ultherapy is safe and effective, ideal for both women and men, with over 1,750,000 treatments performed worldwide. The safety and efficacy of Ultherapy have been established through published clinical studies.
With almost 2 million Ultherapy treatments performed globally, this treatment has been rigorously tested for safety. Its efficacy has also been proven through clinical studies that have been published in many journals.
How many Ultherapy® Prime treatments will I need?
In general, one treatment is usually required for most patients. However, some patients may benefit from additional treatments based on the degree of skin laxity and how our body responds to ultrasound energy and the collagen-building process.
Future Ultherapy® Prime treatments can help keep up with our body's natural ageing process. Because this treatment boosts collagen and elastin production, it will lift and firm skin so you can look younger as you grow older.
Can Ultherapy® Prime be used to lift the brows?
Yes, clinical studies have proven that its micro-focused high-intensity focused ultrasound with real-time imaging technology can lift an average of 1.7mm to 1.99 mm[2] of the brow in just one session.
Can Ultherapy® Pime improve the appearance of lines and wrinkles on the décolletage?
Many patients are now paying more attention to the often neglected area, the decolletage. Crepey skin on the decolletage is caused by skin laxity due to a decrease in collagen production in the body, dry skin and sun-damaged skin.
This results in lines and wrinkles on the chest area that lends an aged appearance. To revert this, Ultherapy® has shown visible and significant results within 90 to 180 days post-treatment[3, 4].
What side effects can I expect from Ultherapy® Prime?
Your skin might appear flushed, but the redness should disappear in a few hours. Some patients may experience tingling, tenderness to the touch, or even slight swelling, bruising, or numbness, but these side effects are temporary.
Ultherapy Prime has an excellent safety profile with over 2 million treatments performed globally and more than 50 clinical studies supporting its efficacy and safety."
Can Ultherapy® Prime replace a facelift?
Ultherapy® Prime is a non-surgical face lifting procedure that treats the deep skin layers to stimulate collagen production, tightening your skin from within.
Collagen is an essential scaffold protein that provides the skin's foundation of elasticity and firmness.
The quality of your skin's collagen plays a critical role in achieving the desired results. It is not the same treatment as a surgical facelift.
Can we consume or apply protein/collagen to replace the collagen loss in our skin?
Unfortunately, collagen soup, powder, supplements, drinks, or skincare products do absolutely nothing to your skin. Our bodies cannot absorb the collagen we eat or apply to replenish the lost collagen in our skin as the molecules are too big to be able to enter our bloodstream or skin.
Every protein we eat needs to be broken down into amino acids before our body can absorb and benefit from its goodness. But we cannot know how much of the absorbed amino acid goes directly to the skin.
Does Ultherapy® Prime melt fat?
No, Ultherapy® Prime does not melt fat.
Is HIFU the same as Ultherapy®?
Many people think that High-Intensity Focused Ultrasound (HIFU) and Ultherapy are the same treatment, but it is not true.
Unlike HIFU, Ultherapy has the added benefit of real-time imaging and Micro focused Ultrasound technology.
Additionally, HIFU and Ultherapy use different types of ultrasound beams.
How to spot a fake Ultherapy® Prime machine?
Before you book your consultation appointment date, please ensure and note that:
Beware of cheap deals and discounted treatments too! If your treatment is heavily discounted, question its authenticity. Some things are just too good to be true!
How long do Ultherapy® Prime results last?
Ultherapy® Prime generally lasts for about 1 to 1.5 years. Initial skin lifting results can be observed immediately post-treatment, with continued optimal results developing over 2 to 3 months as new collagen continues to grow.
Some patients have reported witnessing continued improvement up to half a year.
Doctors recommend annual maintenance treatments to maintain the taut appearance and counteract the effects of natural ageing. Individual results vary based on several factors such as lifestyle, age, natural skin condition and collagen production capacity.
Is Ultherapy Prime painful?
The ultrasound energy will penetrate deep into our skin to heat the tissue between 60 to 70°c. Some patients describe the experience as a warm sensation and some shared that the tingling feeling may be slightly uncomfortable.
However, the discomfort is well-tolerated and will subside quite immediately post-treatment. Numbing cream and oral painkillers can be provided.
How long does an Ultherapy® Prime treatment take?
Ultherapy Prime's enhanced efficiency means treatments are approximately 20% faster than legacy systems.
The session usually lasts from 30 to 90 minutes, depending on the area.
Can Ultherapy Prime be combined with other treatments?
At Amaris B. Clinic, Dr Ivan Puah may recommend combining Ultherapy® Prime with our SkinHydra Booster or UltRA+ Lift for comprehensive facial rejuvenation. The treatment plan is customised based on individual assessment.
Reference
[1] Pavicic T, Ballard Jr, Bykovskaya T, Corduff N, Hirano C, Park Jy, Saromytskaya A, Sevi J, Vasconcelos S. Micro Focused Ultrasound With Visualisation: Consensus On Safety And Review Of Energy-Based Devices. J Cosmet Dermatol. 2022 Feb;21(2):636-647. DOI: 10.1111/jocd.14666. Epub 2021 Dec 24. PMID: 34951735.
[2] Alam M, White Le, Martin N, Witherspoon J, Yoo S, West Dp. Ultrasound Tightening Of Facial And Neck Skin: A Rater-Blinded Prospective Cohort Study. J Am Acad Dermatol. 2010 Feb;62(2):262-9. DOI: 10.1016/j.jaad.2009.06.039. PMID: 20115948.
[3] Fabi Sg, Massaki A, Eimpunth S, Pogoda J, Goldman Mp. Evaluation Of Micro-Focused Ultrasound With Visualisation For Lifting, Tightening, And Wrinkle Reduction Of The Décolletage. J Am Acad Dermatol. 2013 Dec;69(6):965-71. DOI: 10.1016/j.jaad.2013.06.045. Epub 2013 Sep 17. PMID: 24054759.
[4] Fabi Sg, Goldman Mp, Dayan Sh, Gold Mh, Kilmer Sl, Hornfeldt Cs. A Prospective Multicenter Pilot Study Of The Safety And Efficacy Of Micro-Focused Ultrasound With Visualisation For Improving Lines And Wrinkles Of The Décolleté. Dermatol Surg. 2015 Mar;41(3):327-35. DOI: 10.1097/DSS.0000000000000322. PMID: 25705947.
[5] Levine, J., McCarthy, A. D., Pavicic, T., Green, J., & Akers, J. (2025). Comparative Ultrasound Visualization Analysis Between Ultherapy® and Ultherapy PRIME and Survey and Clinical Case Results from the Ultherapy PRIME Early Experience Program.
[6] Panithaporn, D. (2025). Pain, Erythema, and Edema After Facial Lifting With Ultherapy Prime or Ultherapy Legacy—A Survey Study. Journal of Cosmetic Dermatology, 24(9), e70467.
[7] Levine, J., McCarthy, A. D., Pavicic, T., Green, J., & Akers, J. (2025). Comparative Ultrasound Visualization Analysis Between Ultherapy® and Ultherapy PRIME and Survey and Clinical Case Results from the Ultherapy PRIME Early Experience Program.

Dr Ivan Puah's novel 360° Glandular Tissue Dissection (360°GTD®) technique minimises surgical invasiveness while achieving cosmetic results.


Dr Ivan Puah's novel 360° Glandular Tissue Dissection (360°GTD®) technique minimises surgical invasiveness while achieving cosmetic results.


Dr Ivan Puah's novel 360° Glandular Tissue Dissection (360°GTD®) technique minimises surgical invasiveness while achieving cosmetic results.
